Safety and Storage
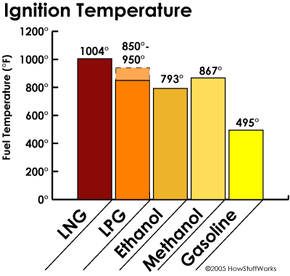
LP gases are actually quite safe in comparison with other fuels. Propane has a high ignition temperature, about 850-950 F (450-510 C), compared to about 495 F (257 C) for gasoline. This makes it less likely to ignite spontaneously.
Also, the tanks used to store propane are stronger than gasoline tanks because of the pressure needed to keep the propane in liquid form. This makes on-board propane storage safer than a typical gas tank -- it is more resistant to rupturing in the event of a collision. Special safety valves and cut-offs increase the safety factor.
Advertisement
To store LP gas safely, there are some guidelines. It's important to remember that a propane tank is never really empty. When the tank is filled, most of the propane is under enough pressure that it is in liquid form. But at safe pressures, not all of the propane is liquefied -- a small amount is in gas form, filling up the rest of the space in the tank. As more propane is used, the pressure decreases, leaving less propane in liquefied form and more propane gas filling up the remaining space.
Tanks should only be filled to roughly 80 percent capacity. Changes in temperature can change the pressure inside the tank. If you were to fill your tank to 100 percent on a cool, cloudy day, and then leave your car out in the sun the next day, the increase in temperature would cause in increase in pressure within the tank that could cause it to fail without that 20 percent headroom.
For more information on LP Gas and related topics, check out the links on the following page.
Related HowStuffWorks Articles
More Great Links
- World LP Gas Association: About LP Gas
- Four Wheeler Magazine: Crawl This Way - an rock-crawling truck converted to propane so it will "run upside down"
- Alternative Fuel Station Locator
- Propane Gas Association of Canada
- Ford Scorpio: A Guide to LPG Conversion
Sources
- Ford Scorpio: LPG Conv (2.0 16V) http://www.fordscorpio.co.uk/lpgconversion.htm
- Global Autogas Industry Network http://www.worldlpgas.com/gain/index.php
- World LP Gas Association: About LP Gas http://www.worldlpgas.com/mainpages/aboutlpgas/distribution.php
- NPGA.org: Snelling Discovery http://www.npga.org/files/public/Snelling_Discovery_1960s.pdf
- BTU Content Comparisons of Common Fuels http://www.cbbqa.com/grilling/Btuchart.html
- Propane Gas Association of Canada http://www.propanegas.ca/files/Propane%20properties.pdf
- Leicester Auto & Marine Gas Services http://www.leicesterautogas.co.uk/lpg_sub.htm
- Do-It-Yourself Automotive LPG Conversion http://www.wps.com/LPG/LPG-book-final.html
- Calor: LPG Conversion http://www.lpg-vehicles.co.uk/lpg_conversions/lpg_vehicle_conversion.htm
- Turner, CC. The LP Gas Man's Encyclopedia of Methods and Equipment. Moore Gas, 1955.
- The Compressed Gas Association. Handbook of Compressed Gases. Van Nostrand Reinhold, 1990. 0-442-21881-8.